Wright Place, Wright Time? Ortho Specialist's Focus Is Paying Off
Executive Summary
In reinventing itself as a niche specialist in the orthopedic market, Wright Medical is putting efforts into maintaining leadership in the upper and lower extremities and building on its biologics strengths – a sales platform that was reinforced highly synergistically on the acquisition of Tornier. But for however long Wright Medical can remain independent, how does it balance value-based health care with strategic development plans?
- Within orthopedics, Wright Medical caused a stir when it restructured away from hips and knees in 2013 and firmly re-set its strategy on extremities and biologics after buying Tornier in 2015.
- That added sales bulk, but as a medium-sized player in a $47 billion orthopedic market, it will never challenge the big five for sheer size. In pursuing niche opportunities representing an $8 billion global market, it is focusing on its strengths and is arguably in the top tier group in matters of innovation.
- So what? Fast-growing Wright Medical has become big enough to write its own script and is putting strategy and innovation issues front and center. But it also has unique market access and outreach techniques that give it a stake in the value-based and outcomes-oriented industry that looks beyond the technologies per se.
The M&A question is never far away for medtech companies that successfully restructure, set ambitious but achievable goals and whose portfolios and markets are relatively easily defined. Thus, device industry commentators have recently been prone to couple upper/lower extremities and biologics manufacturer Wright Medical Group NV, newly augmented by its 2015 Tornier NVacquisition, with suitors as the combined group continues to show robust financial performances.
Add to the mix the contention in some quarters that president and CEO Bob Palmisano has had an M&A agenda in mind since taking over in 2011, and there is little to dissuade observers from picturing a Smith & Nephew PLC(no overlaps) making a bid, a Stryker Corp. or a Zimmer Biomet Holdings Inc.(competition issues likely) showing interest, or even spinal specialist Medtronic PLC looking to build out in ortho while avoiding taking on the hip and knee giants.
It's possible, and maybe even probable. But for now, Wright Medical has more immediate preoccupations, as it continues to integrate Tornier, restructure and augment its sales force and factor in innovations. In the shift away from fee-for-service and toward outcomes-based health care delivery, Wright's management will also be mindful that larger rivals in the ortho space are thinking beyond their technologies per se and introducing new approaches to the market to remain competitive.
Strategy Execution And Innovation Top The Agenda
But that too is a lesser priority at Wright Medical for the time being, as the company focuses ever more on current and prospective business strategies, and in doing so is gaining an increasingly strong position in the market. Speaking to In Vivo, senior vice president and chief communications officer Julie Tracy says Wright Medical is concentrating on its fast-growing orthopedic markets, "where technological innovation is making a difference." As such the company is more focused on growth and execution of the strategy, and hasn't made any public comment on value-based themes. (See sidebar, "Pace Toward Value-Based Health Care.")
Building The New Wright Medical
Meanwhile, Wright Medical is determined to play to its strengths. Certainly, the group under Palmisano is putting a lot of effort into building out a portfolio that underlines the areas of specialization of the "New Wright Medical," which began taking shape when Wright decided to sell its OrthoRecon hip and knee implants business to MicroPort Scientific Corp. That was in summer 2013. In 2017, it sold the Tornier hip and knee business to Corin Group PLC. It has become "a completely refocused company," in the words of Tracy. [See Deal][See Deal]
Speaking at the Jefferies Healthcare Conference in London last year, Tracy explained that, post-transformation, Wright Medical is active in the two highest-growth areas of orthopedics: extremities and biologics. In fact, since fall 2016 at least, Wright has been claiming that its markets have been growing at twice the market rate. This is an $8 billion market combined. (See Exhibit 1.)
Within these segments, Wright holds a leading position in upper (shoulder, wrist and hand) and lower (foot and ankle) extremities. It believes it is the number-one player in the total ankle replacement (TAR) and is the number-two player in the US shoulder replacement market, a position being bolstered by its new reverse glenoid product, PerFORM Reversed Glenoid, launched March 2017. In Augment Bone Graft, it also claims to have the fastest-growing biological product in orthopedics.
"We're not the largest, but we are the fastest-growing and the most focused. In the shoulder market, we're number two behind Zimmer-Biomet, which also has a foot and ankle business," Tracy asserts. Others competing in Wright Medical's foot and ankle space include Stryker and a host of smaller companies. "But we believe we're a technological leader in both shoulder and foot and ankle – and by a wide margin in TAR," adds Tracy.
Exhibit 1
The New Wright Medical's Focus Markets
Wright Medical says its chosen product focus areas address markets with a combined value of $8 billion in 2018. It believes the dominance of the global $47.3 billion orthopedic industry (2016 figure) by five multinationals allows mid-sized companies a chance to focus on the less contested, higher-growth sectors.
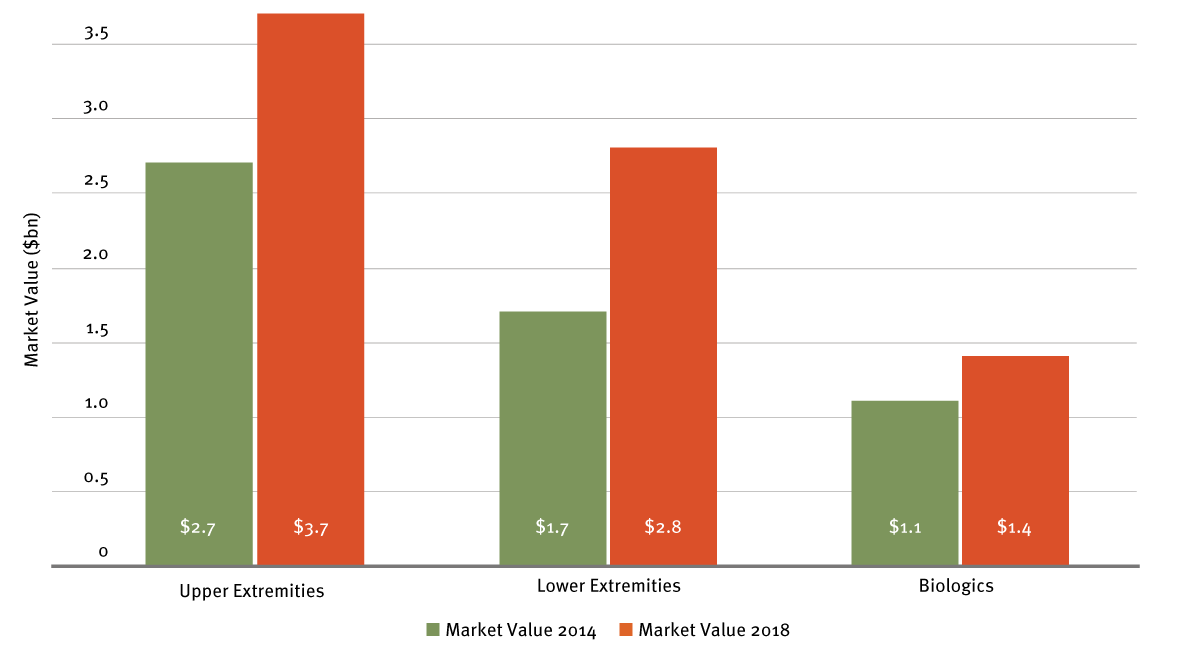
Note: Augment Bone Graft addresses a $300 million market opportunity in US hind foot fusion and ankle fusion; Simpliciti is a player in a $200 to $250 million market; the TAR (total ankle replacement) market could in time be valued up to $400 million to $500 million globally.
Wright Medical
In the fiscal year ended December 25, 2016, Wright Medical had net sales of $690.4 million and a net loss from continuing operations of $164.9 million. (See Exhibit 2.)
Exhibit 2
Group Sales And Segments Breakdown
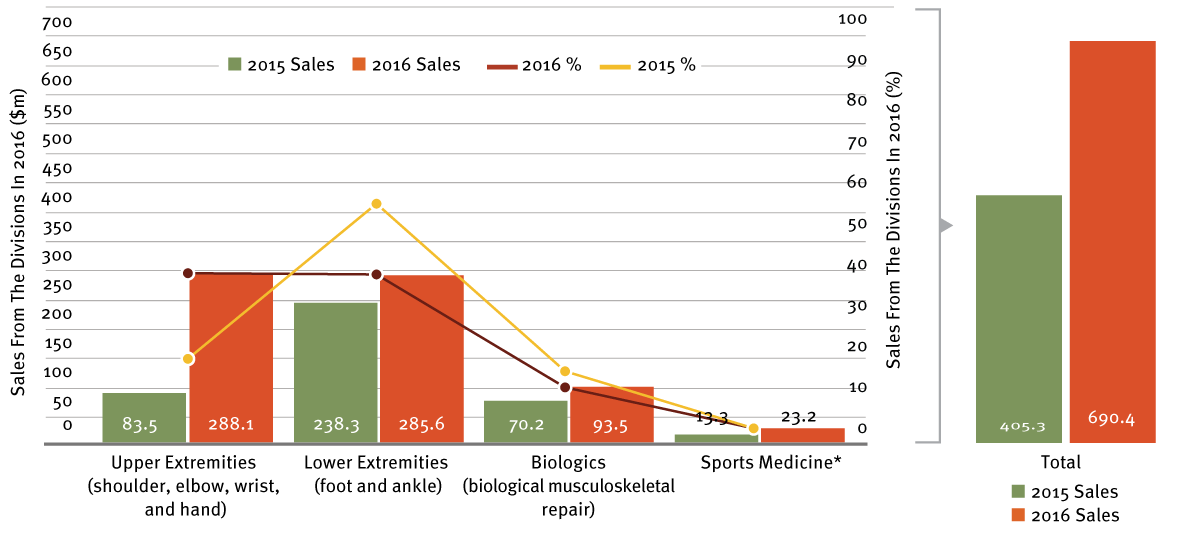
*Includes products used across several anatomical sites to mechanically repair tissue-to-tissue or tissue-to-bone injuries; complements the upper and lower extremities product portfolios.
Wright Medical
Wisdom Of Adding Tornier
Tornier doubled the size of the company when the merger deal closed in October 2015. (Also see "Wright Moves More To Extremes With Tornier Purchase Agreement" - Medtech Insight, 28 Oct, 2014.) It was a coming together of two industry segment leaders of equal size, so "the merger made good strategic sense," Tornier exerting upper extremities market dominance and Wright powerful in the lower extremities markets. The deal gave Wright the scale and scope to accelerate along the pathway to profitability – the wisdom being that this was a pathway neither company would have been able to pursue as fast, had they remained independent. [See Deal]
The discussions started in early 2014. It is now almost two years since the merger, and the "integration has gone extremely well," Tracy says. Wright has pursued the global merger of all sales teams, and completed an upper extremities integration project ahead of schedule. It reported in early 2017 that cost synergies started to materialize earlier than anticipated, and that revenue dis-synergies have played out better than expected. It has also spoken of a materially improved balance sheet.
"Never say never – we're always on the lookout for interesting, unique, differentiated technologies that are complementary with what we do." – Julie Tracy on Wright's readiness for more M&A
It was a combination of strengths that delivered a well-defined and transparent company. "This was a big merger for us and there are no obvious product gaps," says Tracy. Indeed, there is no need to acquire upper and lower technologies, but she cautions, "That said, you never say never, and we're always on the lookout for interesting, unique, differentiated technologies that are complementary with what we do in the areas we are focused on."
Palmisano has recently commented in results sessions that Tornier had a very well-developed international and back office infrastructure. The combined group has manufacturing plants at three global locations: Montbonnot, France; Ireland (Macroom, Cork); and at Arlington, TN. Sales, marketing and R&D premises in Memphis, TN, serve as the US headquarters, whereas the group's global corporate headquarters are located in Amsterdam, the Netherlands.
Wright expects to complete the merger integration and realize all its targeted cost synergies over the course of the next two years.
The restructuring around Tornier was preceded by several smaller extremity company M&A plays by Wright Medical, including, in January 2013, the purchase of UK company WG Healthcare Ltd.; and in 2014 the US manufacturers Solana Surgical LLCin January ($48 million cash and $41.4 million common stock), and OrthoPro LLC in February ($32.5 million). [See Deal][See Deal]
Differentiators Make A Difference
The positive news at the end of 2016 going into 2017 came thick and fast, with Wright claiming that the upper extremities market was growing at 8% to 9%, lower extremities at 8% to 10% and biologics at the rate of 5% to 6%. When a company is in high-growth markets, "differentiators matter," says Tracy, who believes Wright Medical sets itself apart in several ways due to its segment leaderships in the US, its international markets coverage, its stream of innovations and its full R&D pipeline. It also puts a strong emphasis on medical education, she says.
A big focus of the company is on these differentiators. "These are areas where we have unique and competitive advantages and where we can make a difference with customers."
Even with the merger, the US remains the dominant territory, delivering roughly 70% of Wright's sales.
Even with the merger, the US remains the dominant sales territory, delivering roughly 70% of Wright's sales. The group has direct sales teams in the larger international markets, such as the US, Germany, France and the UK, where it has a "presence of a decent size." It is also strong In Brazil, the rest of Latin America, China, Canada and Australia. It markets its products in over 50 countries worldwide. But its presence is selective: "We're not going to be in every single international market," says Tracy.
Value-Based Innovation – Wright Medical Style
Wright Medical operates a range of innovative initiatives that go beyond the technology and serve the clients' own needs. These include:
Enabling Technologies
These are part of the group's strategy and are driving growth in all areas. Blueprint and Prophecy, used in TAR, are enabling technologies that help physicians pre-plan procedures, thereby saving time in the OR. According to Tracy, published data on Prophecy shows it can save as much as 45 minutes in the OR. It is also designed to make the procedures easier and more predictable – with better alignment – all the elements that are going to produce better patient outcomes in the long term.
Predictability
Wright Medical is also continuing to add efficiency and predictability to its operations by expanding its Inventory Hub Network. This has been operating for several years for lower extremities. "We have an active plan today to move the upper extremities inventory into those same hubs, as a result of the merger with Tornier," notes Tracy.
This unique hub network will prospectively include around 20 distribution points in the US. The system is conceived to replace the individual distribution systems run by each rep or each distributor in the US, and to enable better control of inventory. It frees up reps' time so they can focus on sales, and gives more control and visibility over the performance of the field inventory and instrument sets. Experience has shown that this leads to a reduction in field inventory days on hand.
The hubs also deliver more information, allowing Wright to be data driven about inventory and instrument needs.
Vital Few Initiatives
The hub network is one of Wright's "Vital Few" initiatives for 2017. These are initiatives that can "move the needle." They are intended to be foci of excellence on selected areas, compared with doing a wide sweep on many areas, maybe imperfectly. "We survey our customers and staff twice a year, and use that data to help us determine what projects are needed," says Tracy.
Just-In-Time Instrument Use
Another Vital Few initiative is a drive to reduce the amount of inventory delivered for surgery. Wright has identified improvements in communication and in ways of interacting with surgeons to ensure that delivery and use practices are optimized. This can reduce the number of kits delivered into surgery by roughly 30%.
"It's not typical in the ortho market to see some of the things that we're working on. Reps will bring in inventory for a particular procedure, and much of it never gets moved," says Tracy. "To the extent that we can predict what is needed for a particular procedure, we can deliver that alone to the right place at the right time."
"The markets we're in are still very early and are where innovation matters in terms of product development." – Julie Tracy
Focus On Innovation
The above may be perceived as Wright Medical's approach to delivering value to its customers beyond mere innovative products. But for Wright, there are rich seams of innovation yet to exploit. "The markets we're in are still very early and are where innovation matters – in terms of product development," says Tracy. "These are not mature markets – unlike hip and knee, where there are a lot of players. Here, innovation is still an important piece and is expanding the market. We are differentiating the technologies."
The "step-down" in value concept – whereby a company drives two separate brands of a similar technology for different markets with different levels of purchasing power – is not a major part of the Wright Medical strategy. (Also see "Medtech Value Brands: Stepping Down May Boost The Top Line" - In Vivo, 21 Jun, 2017.) In the days before Wright Medical's 2013 sale of its OrthoRecon hip and knee business to MicroPort Medical BV/MicroPort Scientific Corp. for $290 million, the group had been pursuing the innovative approach of the rep-less ortho model.
That was when Palmisano crystalized the notion that Wright would become a smaller, high-growth extremities company with breakthrough biologics opportunities, focusing on improving sales productivity, and extending the global reach and penetration of products in key international markets. The plan extended to registering and developing the market for the Augment Bone Graft – to create "significant shareholder value.” (See sidebar, "Augment Bone Graft Biologic – Fastest-Growing In The Portfolio.")
When Wright divested Tornier's small hip and knee business to Corin for €29.7 million ($32.8 million) in cash, it effectively exited the large joints business for a second time. These products were largely marketed in France and other European countries and contributed about $41 million in sales in 2015.
Extremities Innovations And Products In R&D
Wright Medical is in bullish mode right now, and claims to be at the front end of new product cycles in all three of its target areas (with 180 products in upper and lower extremities and 20 in biologics). It spent $50.5 million on R&D in 2016 on its core areas. Some of its recent breakthroughs include:
Lower Extremities
- Infinity Total Ankle System. The third-generation rollout continues. Combining this with Prophecy and Inbone makes Wright Medical "a clear leader" in the total ankle replacement (TAR) market. The TAR-enabling technology (Prophecy) is a key element; with this solution, a CT scan, surgical treatment plans and surgical cutting guides are provided ahead of the procedure. Using Infinity and Prophecy can cut up to an hour off surgery times. "Only approximately 15% of patients who are eligible with end-stage ankle arthritis are getting TAR today," the group claims.
- Salvation Limb Salvage System. This is claimed to be the first comprehensive solution to treating Charcot arthropathy and to delivering advanced mid-foot reconstruction: where the patient is in danger of losing the foot, this product allows for a recon procedure.
- Invision Revision Ankle System. Anticipated launch third-quarter 2017.
- Ortholoc 3Di Ankle Fracture LP System. Anticipated launch fourth-quarter 2017.
Upper Extremities
- Simpliciti Shoulder System. This is already a year into rollout. It is said to be good for younger patients with good quality bone. It sells at a 50% to 60% premium to traditional anatomical shoulders.
- Blueprint 3D Planning. Being rolled out in stages, it is a software to help physicians plan their cases, try different technologies on a computer without actually implanting them, to see what would work best.
- PerFORM Reversed Glenoid. Launched in March 2017, it is an implant with porous metal fixation specifically shaped for treating patients with difficult glenoid anatomy. It is expected to be a "game-changing product," as more of the US market seems to be moving toward reverse shoulders, a trend already seen in Europe.
Big US Boost For TAR Reimbursement?
The US Centers for Medicare & Medicaid Services is also proposing a major shift in payment rates for primary TAR procedures. If approved, US payment rates would change as of October 1, 2017. The impact would be a $7,000 increase in all TAR procedures – from roughly $12,000 to $19,000 for 2018. "We've been advocating several years for higher reimbursement for TAR, and it's terrific that CMS is now proposing it," says Tracy. The procedure takes longer than a total hip or total knee replacement.
Wright thinks TAR is significantly underpenetrated and could grow by four to five times its current size to a market of some $400 million to $500 million globally.
It is not known how private payers would be affected, but their procedures are typically covered at higher rates than under Medicare payments. This change would support continued conversion from fusion to TAR, says Wright, which thinks that TAR is significantly underpenetrated and could grow by four to five times its current size to a market of some $400 million to $500 million globally.
When Wright commissioned a consultancy to look at the TAR market, it found that up to 90% of US ankle fusion patients are candidates for TAR. The generic technology had a suboptimal start in the early days, when doctors were trying to use a product that did not work as well as it should. But the new products, such as Infinity, are much easier to use, says Palmisano. He adds that, "As the market gets bigger and doctors get more comfortable with the products and our training even gets better, so we see that opportunity."
It is estimated that over 50% of US TAR procedures in 2016 used Wright products, and the group's full support of the market is seen as an important factor in this – with Inbone Total Ankle, Infinity Total Ankle and Prophecy patient-specific guides already launched, and Invision Revision Ankle coming out in the third quarter of 2017. The year 2016 was a record one for Wright Medical, both for annual revenue for total ankles and for the total number of cases performed.
Final Thoughts – Current Year And Beyond
Now as a group "of size," Wright Medical is pressing to achieve mid-teens top-line growth, while over a three-year period delivering $45 million to $50 million of cost synergies as a result of the Tornier merger. Recent guidance anticipates Wright Medical net sales for 2017 of $755 million to $765 million, assuming a two percentage-point negative impact from foreign currency exchange rates. That would result in net sales growth of 12% to 14%, excluding the impact of revenue dis-synergies in constant currency. New product launches and sales force expansion are expected to drive faster growth in the second half of 2017.
The outlook for full-year 2017 non-GAAP-adjusted EBITDA from continuing operations is $78.5 million to $85.5 million. Other performance targets include gross margins in the high 70% range, and a 20% adjusted EBITDA margin in three to four years' time.
In the background, the litigation concerning failure of metal-on-metal hip implants is ongoing. Most recently, the US 11th Circuit Court of Appeals ruled to uphold a $2.1 million verdict in favor of a woman who says she was injured by the company's Conserve artificial hip. (Also see "$2.1m Verdict Against Wright Medical Stands" - Medtech Insight, 27 Mar, 2017.) But Wright was able to reach a Master Settlement Agreement in November 2016 that addressed some 85% of the known revision cases. The parties agreed to settle 1,292 specifically identified Conserve, Dynasty and Lineage claims for an amount of $240 million. Wright has now reached agreement with three of the six product liability insurers.
Despite the many distractions for Wright Medical at present, the group is sticking to a plan. Tracy says, "We're focused on delivering shareholder value, finishing the integration and building a company that's a leader in the markets that we've chosen to be in." It's a strong and clear message from a group that has transformed itself, doubled in size, is not afraid to focus on self-identified client needs and appears to have the route ahead nicely mapped out.
