In Vivo's Top 10 For 2017
Executive Summary
Our 10 most-read thought leadership pieces of last year. Not surprisingly, our looks at pricing, reimbursement and patient access scored well. Readers also responded to our deep dives into technology innovation and our conversations with industry leaders. (Free article.)
Leadership
Bill Looney's conversation with Pfizer's Peter Honig was our top in-depth Q&A of 2017, followed by Chris Morrison's interview with Bayer's Robert LaCaze.
Pfizer’s Safe House: Peter Honig Builds A Regulatory Model For The Future
Under Honig, Pfizer's worldwide safety and regulatory division has become an unexpected hotbed of process innovation – and the only operational unit at Pfizer that monitors every stage of the drug product life-cycle, from discovery to senescence.
Inside Bayer Oncology: An Interview With Robert LaCaze
Bayer AG may more often make headlines for its deal-making in consumer health and agriculture, but the massive German conglomerate sees oncology drugs as a key engine of future success within its high-growth pharmaceuticals business. A Q&A with the EVP of Bayer's new Oncology Strategic Business Unit.
Market Access
In Vivo's readers devoured everything we wrote on pricing, reimbursement and market access in 2017 These three articles made it into our Top 10.
US Biosimilars: Busting Blockbusters Or Managing Obsolescence?
As a new generation of biosimilars challenge some of pharma’s biggest-selling products, John Hodgson looks at how the originator blockbuster might fare in the face of competition. Experience in Europe, which predates that in the US by about a decade, provides a number of crucial pointers to outcomes and defensive strategies.
New Payment And Financing Models For Curative Regenerative Medicines
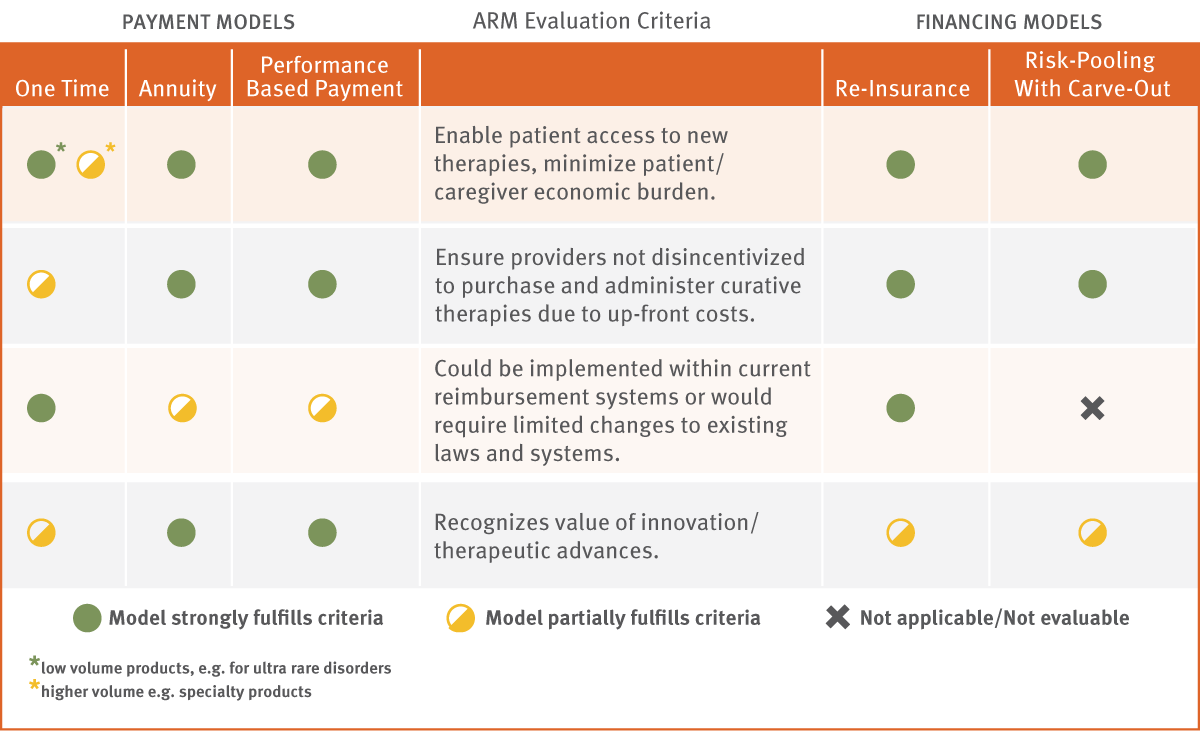
Ranking the Models
Second in a series of white papers written for In Vivo by the Alliance for Regenerative Medicine. Several reimbursement and financing models have been proposed to address the potential uncertainty and economic disincentives that may be associated with curative therapies. Implementing them will be critical to ensuring patient access to those medicines and continued innovation in the field.
To Get Pricing Right, Pharma Must Understand Payer Behavior, Trump Or No Trump
Shortly after his presidency began, In Vivo contributor Melanie Senior asserted that whatever President Trump says today or does tomorrow, pressures on drug pricing aren’t likely to ease any time soon. Biopharma firms need to devise and design appropriate pricing and commercial strategies for payers, a group of customers that don’t all care about the same things and, in some cases, whose needs may be diametrically opposed.
Technology Innovation
Our close looks at antibody drug conjugates, artificial intelligence and diagnostics resonated with readers in 2017.
Can Artificial Intelligence Help Find Better Medicines?
Melanie Senior argues that AI might help streamline some components of drug discovery, like rapid screening of virtual molecules, and providing a wider selection of viable hits, but it is not yet the answer to pharma's R&D productivity problem. Moreover, its impact will be felt as just one part of the wider data analytics revolution that pharma must embrace to remain competitive.
Companion Diagnostics: The Expanding Reach Of Personalized Medicine
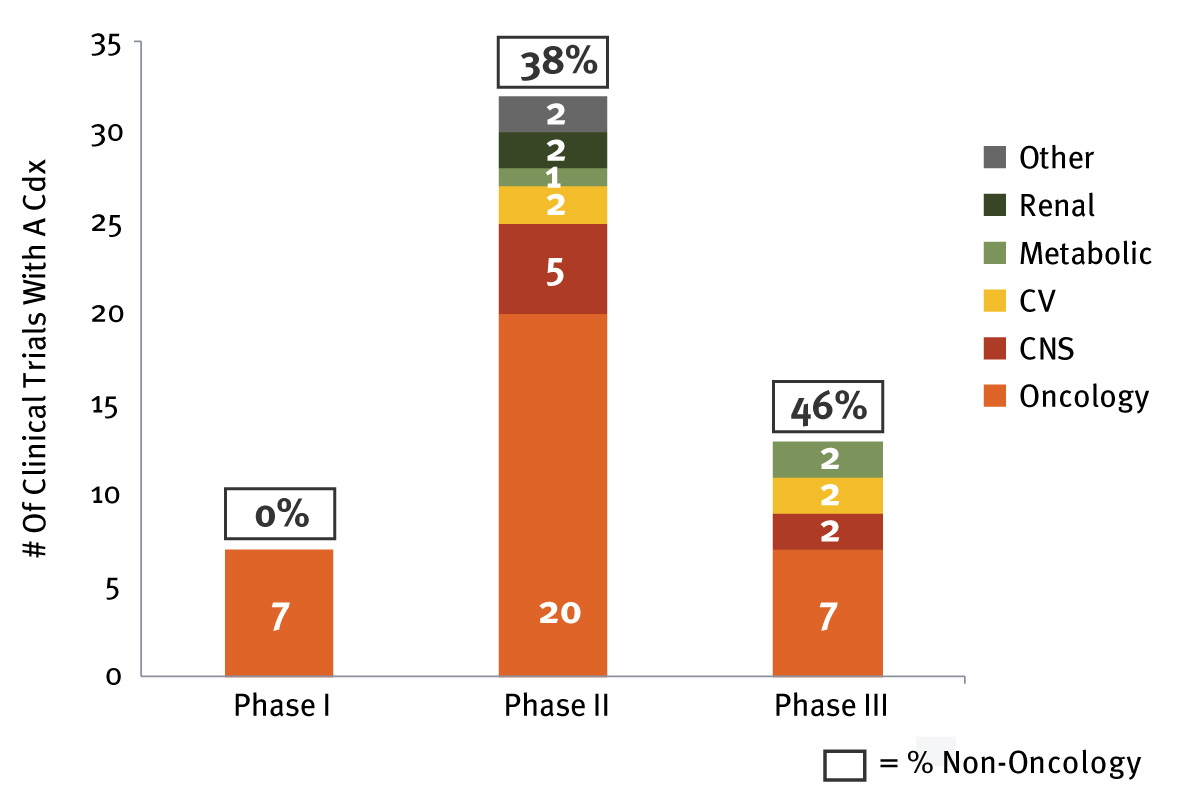
Companion Diagnostics Pipeline
Personalized medicine is becoming the hallmark of care in oncology, but its use is also increasing in other therapeutic areas including inflammation, respiratory, infectious diseases and central nervous system disorders, as scientific understanding of these diseases advances. inVentiv Health notes that the expansion of companion diagnostics beyond oncology has impacts on deal-making, clinical practice and the R&D pipeline.
Cancer’s Next-Gen Smart Bomb: Who Will Be First To Weaponize?
Advances in antibody drug conjugates, if realized in clinical trials, will provide patients with one of the first tangible benefits of precision medicine. ADCs may also answer the question that has dogged cancer treatment for decades: how to destroy a tumor without killing the patient in the process. Bill Looney profiles two biotechs in the space: Ambrx and Sutro Biopharma.
Liquid Biopsy On Course To Transform Cancer Management
Peter Charlish surveys the liquid biopsy market. The technology is playing an increasing role in drug development, but it will be a hurdle to move them into clinical practice. Younger physicians are likely to be early adopters, whereas older physicians will likely cling to more traditional techniques such as tissue biopsy.
Market Intelligence
This infographic was the centerpiece of our March 2017 themed issue on personalized medicine.
Personalized Medicine: An Infographic
There were 132 personalized medicines on the market in 2016, compared with just five in in 2008, according to the Personalized Medicine Coalition. And 27% of new molecular entities approved by the FDA in 2016 can be can be classified as a personalized medicine. That's a big jump from 2005, when personalized medicines accounted for just 5% of NME approvals. A look at some other key numbers in this rapidly growing field.
Thank you for reading this article! Please help us to better help you, by taking our Pharma Feedback survey. Not only will your participation help steer our efforts to continually improve the content and delivery of our products – you’ll also have a chance to win one of four Amazon gift cards valued at $100 (US).


