Fortress Biotech: A Drug Financing Experiment Expands
Executive Summary
Fortress Biotech has established nine subsidiary companies since 2013, each financed differently and each focused on an area of unmet need. Fortress CEO and longtime life sciences investor Lindsay Rosenwald lays out his ambitious plans for the future.
- Fortress emerged from the ashes of Coronado Biosciences in 2013 with a new approach to biotech company creation. It builds companies around promising science and finances them mostly through relatively modest up-front investments. Fortress maintains a consistent revenue stream by charging its subsidiaries annual consulting fees.
- Fortress' subsidiaries cross multiple therapeutic areas, but several are working in cancer, hospital products or rare disease. Rosenwald's long-term vision is for Fortress to become a fully integrated biopharmaceutical company, incorporating R&D and manufacturing.
- So what? Fortress Biotech's approach to company creation is bold and ambitious, but executing on the strategy and bringing medicines to market will be the ultimate mark of success.
"We wanted to do a lot quickly," Fortress Biotech Inc. CEO Lindsay Rosenwald, MD, says over the boardroom table in the company's glass-enclosed top-floor conference room overlooking Manhattan's meatpacking district. The longtime life science investor's idea of doing a lot quickly? Launching nine biotech subsidiaries in less than three years, all under the Fortress umbrella.
Rosenwald has big plans for the future of Fortress too: establishing more subsidiaries, progressing pipeline drugs through clinical development and onto the market, and watching the return on investment come in. Getting drugs to market, generating revenues and profits is what will ultimately determine the success of Rosenwald's Fortress experiment.
Fortress is a start-up/venture investment firm hybrid. It scours medical and academic research labs and pharma's basement laboratories for compelling science and shelved assets, secures financing for the programs, and establishes a new company and leadership team around them. The model is different from a traditional venture firm in that Fortress doesn't always front the cash to fund each company's product development and it maintains each subsidiary under the parent umbrella. The subsidiary structure protects Fortress against substantial future dilution, while providing a consistent revenue stream because subsidiaries pays Fortress an annual $500,000 consulting fee.
"There is no shortage of tremendous product leads. There is no shortage of capital. The shortage is entrepreneurship." – Lindsay Rosenwald
The business model is reminiscent in some ways of Vivek Ramaswamy's Roivant Sciences Ltd., which also invests in overlooked science and establishes subsidiary companies that it retains an interest in. But if Roivant, the firm behind the most successful biotech IPO of 2016, Myovant Sciences Ltd., which raised $218 million and brought in a big pharma backer, Pfizer Inc., has made a cannon ball-sized biotech splash, Fortress is operating under the radar. (Also see "Myovant CEO Says Buoyant IPO Reflects High Interest In Women's Health" - Scrip, 28 Oct, 2016.)
Rosenwald, however, sees an opportunity to front smaller amounts of cash in under-appreciated assets that could still yield substantial returns. "There is no shortage of tremendous product leads. There is no shortage of capital. The shortage is entrepreneurship," he says. "There is just not enough people that have the skill and the desire and the ability to handle that much pain to do this."
Fortress subsidiaries all operate out of the company's central headquarters under which Fortress provides administrative and other assistance, including access to potential financing partners, a business development team and entrepreneurial leadership. Rosenwald's long-term vision for Fortress is for it to become a fully integrated biopharmaceutical company, eventually incorporating R&D and manufacturing.
A Diverse Portfolio Hemmed By Opportunity
The deals Fortress has signed thus far are centered around relatively modest up-front payments by biotech standards, with the deal structures being heavily back-end loaded. Fortress retains an economic interest in each of the companies it launches, either through an equity stake or royalty rights to development programs.
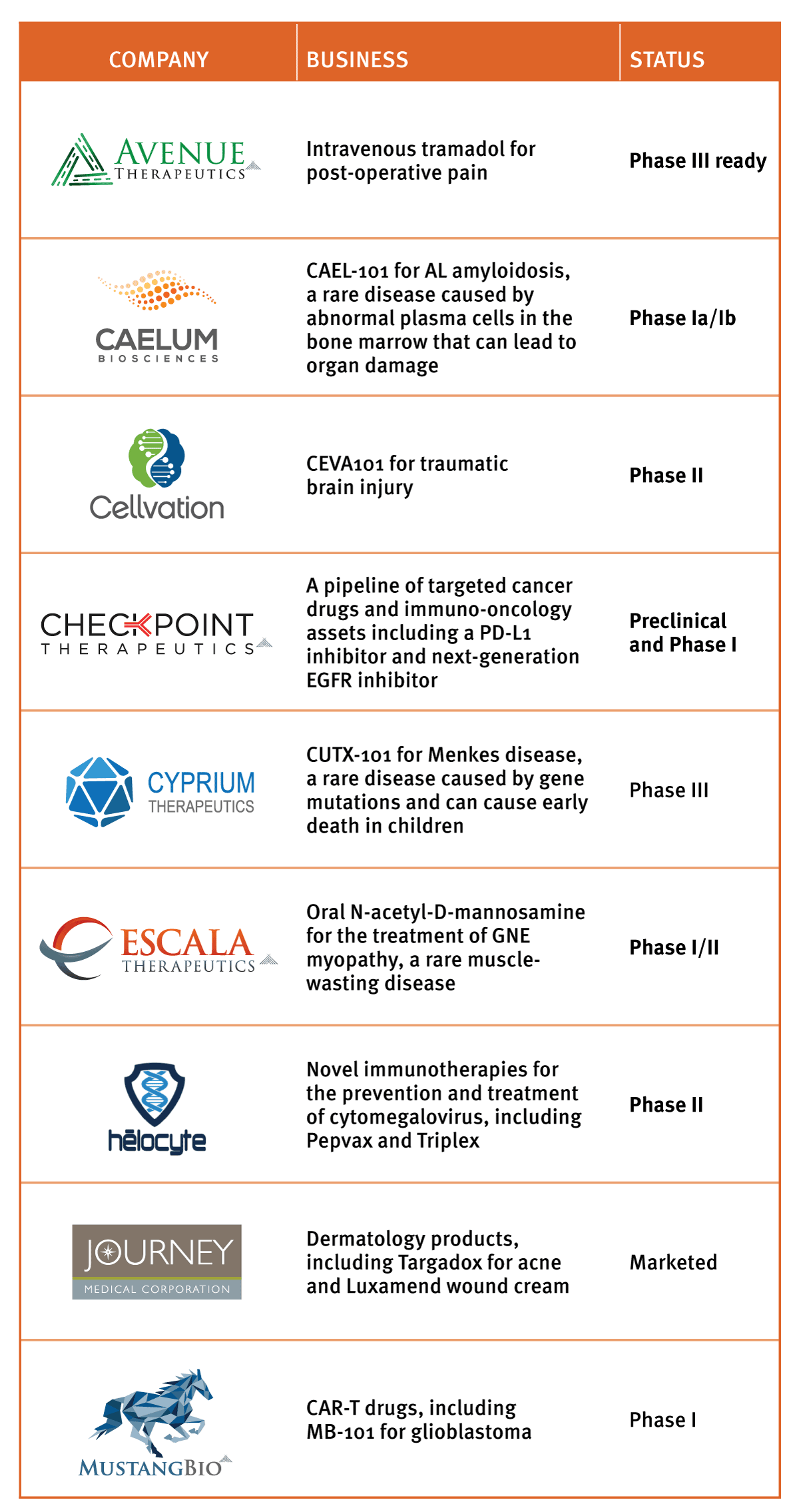
The company's nine subsidiaries cross a range of therapy areas; the only real requirement Rosenwald has when looking for new assets, he says, is that the drug programs address an important unmet medical need. The result is that the Fortress subsidiaries represent a complex patchwork of drugs rather than a focused R&D strategy. Nonetheless, each subsidiary remains therapeutically focused, which helps to narrow the scope within the broader company. (See Exhibit 1.)
"We pick areas where we feel there is a huge unmet medical need, where we think we could have an impact," Rosenwald says. The portfolio includes several subsidiaries working in cancer, such as Mustang Bio Inc., which is developing a chimeric antigen receptor (CAR)-based immunotherapy for brain cancer, and Checkpoint Therapeutics Inc., which is focused on developing targeted cancer drugs and immuno-oncology assets.
Others are working in rare diseases, like Escala Therapeutics Inc. and Helocyte Inc., which is developing therapies for the prevention and treatment of cytomegalovirus. The newest subsidiary, unveiled March 14, is Cyprium Therapeutics Inc., which is focused on treatments for the rare pediatric genetic condition Menkes disease and related copper metabolism disorders. (Also see "Fortress Adds New Rare Disease-Focused Subsidiary" - Scrip, 15 Mar, 2017.)
In pursuit of new assets, Rosenwald has created a business development team of about 20 people, most of whom are working in specific therapeutic areas. "We have somebody just doing pediatric rare diseases, someone doing disease of the liver, someone doing leukemia, degenerative neuro diseases."
"Their job is to build super key opinion leader networks in each of their areas, so that after six months, everyone knows Fortress and Fortress knows everyone in that field," he adds. "That's where you find the really great opportunities in that field. You don't find them at the licensing office at Harvard."
Frank Taffy, who knew Rosenwald from shared time working at the investment firm Paramount Biosciences, where Taffy was VP and general counsel, was one of the first business development executives to join Fortress. He has founded two subsidiaries since 2015, which he now runs as CEO, Helocyte and Cellvation Inc., which is developing a new approach to treating traumatic brain injury.
"Lindsay brought me in as an entrepreneur-in-residence with the exclusive objective of identifying programs around which we could form and finance a company," Taffy says. "My search was agnostic in terms of therapeutic area, but ultimately, I learned that focus makes your search more effective. I became interested in glioblastoma and started speaking to a number of experts that led me to cytomegalovirus."
Fortress has been ramping up the business development group in the last two years, with the intention of adding one or two new assets each quarter, but as the business development team has grown, existing subsidiaries have matured to the point where some are now considering their own business development initiatives, Rosenwald expects the pace of business development will pick up.
A Company Evolves
As is often the case in biotech, Fortress was born out of a former company, Coronado Biosciences Inc., a public biopharma focused on developing novel immunotherapies for the treatment of autoimmune disease. But Coronado reported disappointing efficacy data on one of its lead assets known as Trichurissuis ova (TSO), for Crohn's disease, in 2013, resulting in a management and strategy shake-up.
Rosenwald, who had founded the company and served as a director since 2009, was appointed chairman and CEO in December 2013 and used the opportunity to re-brand Coronado. He was previously a portfolio manager and partner at the asset management firm Opus Point Partners, and from 1991 to 2008, he was chairman of Paramount BioCapital. Over his career, he helped to establish and capitalize numerous public and private biotechs, some with more success than others.
Rosenwald's best known biotech success story is probably Cougar Biotechnology Inc., the company that developed the prostate cancer drug Zytiga (abiraterone) and was sold in 2009 to Johnson & Johnson for close to $1 billion prior to the drug's approval. (Also see "J&J to buy Cougar for $1 billion" - Scrip, 22 May, 2009.) There have been other checkered experiences over his lengthy career, like Indevus Pharmaceuticals, which saw its weight loss drug Redux withdrawn from the market but later reinvented itself.
A partner of Rosenwald's at Paramount and Opus Point, Michael Weiss also joined him at Fortress. Weiss has been director and co-vice chairman of the board at Fortress since December 2013 and he became EVP, strategic development in January 2014. He was the CEO of Keryx Biopharmaceuticals Inc., which eventually developed the marketed phosphate binder Auryxia (ferric citrate) to control phosphorus serum levels in patients with chronic kidney disease. But the FDA approval came years after Weiss left the company in 2009 following the Phase III failure of another candidate, Sulonex (sulodexide) for diabetic nephropathy, and thus the firm’s pivot to ferric citrate. (Also see "Keryx Suffers Setback As Diabetic Nephropathy Candidate Fails Phase III Study" - Pink Sheet, 10 Mar, 2008.)
Together, Rosenwald and Weiss set about building a very different company from Coronado. They each own 16% in Fortress.
Throughout 2014, the company's strategy evolved, as Rosenwald sought out new technologies and launched six subsidiary companies. In April 2015, Coronado changed its name to Fortress Biotech, and its ticker on NASDAQ to the symbol FBIO, a name the firm says better reflects the company's revised business plan.
Fortress continued the ramp-up in 2015, adding a subsidiary company called Avenue Therapeutics Inc. that is developing one of Fortress' nearest-term commercial opportunities, an intravenous formulation of the opioid analgesic tramadol for pain. The company paid $2 million up front to privately held Irish company Revogenex in exchange for US right to the IV formula, plus milestone payments and royalties on sales of the product, which is now ready to begin Phase III testing after financing is secured.
Exhibit 1
Behind The Walls At Fortress: The Nine Subsiary Companies
Fortress' Cancer Companies Pique Investor Interest
One Fortress subsidiary that is getting attention is Mustang Bio, which is working to develop two CAR therapies that came out of the laboratories of City of Hope. One of the programs, MB-101, is targeting the interleukin-13 α2 receptor, which is overexpressed on the surface of glioblastoma cells. The other is MB-102, a proprietary CAR-T targeting CD123, which is overexpressed on the surface of acute myeloid leukemia cells. Both drugs are in Phase I testing.
Evidence in patients who have been treated with MB-101 has been promising, although it is incredibly early. A case in one study from the Phase I trial in patients with glioblastoma multiforme (GBM) was reported in The New England Journal of Medicine in December. The patient, who had failed tumor resection and prior treatments and had developed tumors in the brain and spinal cord, was given MB-101 into the tumor cavity and later in a novel approach, into the ventricular system in the brain. After the intraventricular treatment, the patient experienced tumor regression on both types of tumors and was able to return to normal life and work. The clinical response lasted 7.5 months after treatment initiation.
The data have generated interest from investors despite the limited nature of the results because of the enormous opportunity for treating GBM. GBM is considered a rare cancer, but deadly, with sparse treatment options. Temozolomide chemotherapy and radiation have been shown to extend survival from 4.5 months to 15 months, according to Mustang. However, the five-year survival rate is less than 10%.
In February, Mustang closed a $94.5 million private placement financing, and the company is continuing the two ongoing Phase I trials and evaluating a potential accelerated approval pathway for MB-101. [See Deal]"Whether we would get accelerated approval or we would have to do a randomized Phase III, I don't know," Rosenwald says. "I know the FDA is very aggressive now about unmet needs in oncology."
Fortress licensed the programs in March 2015 from the cancer center City of Hope and the laboratories of Stephen Forman, director of T Cell Therapeutics Research Laboratory. The company paid $2 million up front for rights to the IP and granted one million in shares of Mustang's stock.[See Deal]
Mustang recently announced the appointment of a new CEO April 24, the experienced CAR-T developer Manuel Litchman, MD. He joins Mustang from Arvinas Inc., where he was CEO, and he previously worked at Novartis AGfor 18 years in oncology R&D. His position was in the big pharma's Cell & Gene Therapies Unit as exec global program head, CTL019, the high-profile CAR-T program Novartis developed under a collaboration with theUniversity of Pennsylvania and which is currently pending at FDA for acute lymphoblastic leukemia in pediatric patients.
Another cancer company formed under Fortress is Checkpoint Therapeutics, which began trading on the OTC market in 2016 through the Form 10 pathway. The company licensed a portfolio of immuno-oncology targeted antibodies from the Dana-Farber Cancer Institute in March 2015, including antibodies targeting PD-L1, GITR and CAIX. Checkpoint paid Dana-Farber $1 million up front plus 500,000 shares of Checkpoint common stock, valued at $32,500. [See Deal]
Dana-Farber is eligible to receive payments of up to an aggregate of $21.5 million for each licensed product upon Checkpoint's successful achievement of certain clinical development, regulatory and commercial sale milestones, as well as up to $60 million in aggregate based on certain sales milestones in addition to royalty payments.
Checkpoint has since completed other deals too, such as a licensing agreement with Teva Pharmaceutical Industries Ltd. in December 2015 in which Checkpoint obtained worldwide licensing rights to a poly ADP-ribose polymerase (PARP) inhibitor in exchange for an up-front fee of $500,000 and up to $220 million in milestone payments. (Also see "Teva Licenses Cephalon PARP Inhibitor To Checkpoint Therapeutics" - Scrip, 8 Jan, 2016.)[See Deal]
In another deal in May 2016, Fortress licensed from Jubilant Biosys Ltd. the rights to compounds inhibiting BRD4, part of the BET pathway, for cancer treatment for $2 million up front. [See Deal]
The company also licensed an epidermal growth factor receptor (EGFR) inhibitor in 2015 from China's NeuPharma Inc. now called CK-101, that is being tested in a Phase I/II clinical trial. [See Deal] The drug is a third-generation EGFR inhibitor that would compete against AstraZeneca PLC's Tagrisso (osimertinib), which is approved for metastatic non-small cell lung cancer with EGFR T790 mutations that have progressed on prior EGFR therapy. "Tagrisso is a really good drug, but they have a whole market to themselves right now, and it is literally a $4 billion to $5 billion market opportunity," according to CEO James Oliviero.
Checkpoint is also working to advance its first immuno-oncology drug into the clinic, a programmed death-ligand 1 (PD-L1) inhibitor, which the company believes it needs to have in the portfolio as a cornerstone therapy to compete in the IO space and to develop novel combinations. "You pretty much have to have a PD-L1 or PD-1 as a base, and then you can layer on top of that, and our goal is to have proprietary combinations in house," Oliviero says. The company aims to develop its PD-L1 inhibitor, CK-301, in combination with the EGFR inhibitor CK-101.
Checkpoint understands full well the competitive dynamics in the field of immuno-oncology, dominated by big pharmas like Bristol-Myers Squibb Co., Merck & Co. Inc., Roche, Pfizer and AstraZeneca, as well as a slew of young biotechs working to develop next-generation drugs.
Nonetheless, the company won't ignore the size of the potential market opportunity for effective IO combinations, and believes that winning an even small slice of the market would be lucrative for a company its size. "When you are talking about PD-1/L1s that have an estimated $40 billion peak sales opportunity in solid tumors, if you get 1% of that, it's $400 million in revenues a year," Oliviero says. "Just being a me-too and pricing it low, you can get market share certainly that is meaningful to a company of our size." The ultimate goal is to do more than that by developing combinations that could extend patients’ survival.
Checkpoint is also interested in bringing in more oncology assets. "Fortress has an incredible BD group," Oliviero says. "We are always watching the new IO therapies coming out, so definitely we are interested in expanding our portfolio."
Mustang and Checkpoint were highlighted in a March 22 research note from JMP Securities initiating coverage of Fortress Biotech. Analyst Michael King initiated coverage of Fortress with a market outperform rating and $11 price target, based on a sum-of-the-parts valuation, focused on Mustang, Checkpoint, Helocyte and Caelum Biosciences Inc. Fortress owns 80% of Caelum, 80% of Helocyte, 39% of Mustang Bio and 37% of Checkpoint, according to the analysis. "Should these subsidiaries realize the potential of the therapeutics under development, we believe the valuations of these FCs will appreciate substantially, as well as Fortress' equity interests therein," King says.
The stock closed at $4.47 on June 12.
JPM Securities analyst Michael King forecasts that Mustang's CAR-T for glioblastoma and Checkpoint's PD-L1 inhibitor/EGFR inhibitor combination could become blockbusters.
"Fortress has assembled a stable of subsidiaries, each of which was funded with modest investment and are making significant progress," King says. Mustang and Checkpoint are two with significant long-term growth potential, he says. He predicted that Mustang's CAR-T for glioblastoma and Checkpoint's PD-L1 inhibitor/EGFR inhibitor combination could become blockbusters. The JPM valuation doesn't include Fortress' other subsidiaries, but King says they could represent significant under-appreciated value.
"Given the substantial commercial potential of the therapeutics under FC development – we believe they could generate in aggregate over $5 billion – we expect Fortress Bio's equity stakes in each of its subsidiaries to contribute significant value in the future."
A Focused Hospital Strategy
Avenue Therapeutics represents a near-term pipeline asset and relatively straightforward commercial opportunity. "Of everything we are doing, this is probably the simplest story, but at the same time it is one of the most compelling stories out there," says Avenue's interim CEO Lucy Lu.
"That could be closest to market," Rosenwald adds of the intravenous version of tramadol under development by Avenue for postoperative pain. Fortress does own one subsidiary, Journey Medical Corp., that markets dermatology products, but otherwise, the subsidiaries are all development-stage. Avenue's asset is ready to begin Phase III testing.
Oral tramadol has been available in the US for more than two decades as Ultram, which was marketed by Johnson & Johnson, but an intravenous version for hospital use was never developed. Meanwhile, IV tramadol is used in many other countries to manage postoperative pain.
J&J didn't show any interest in developing an IV version, says Lu, and there were challenges trying to secure intellectual property protection for the product in the US. However, on May 19, Avenue announced it has received a Notice of Allowance from the US Patent and Trademark Office for a 50-mg version of IV tramadol that helps to secure the company's IP position. "The clinical program will cost us at least $30 million," Lu says. "Without the IP, no one would put in the investment to develop it."
Avenue believes IV tramadol will fill a sweet spot in the hospital market between IV acetaminophen for mild pain and Schedule II opioids that have a high potential for abuse. Tramadol is a Schedule II opioid with a low level of abuse. The hospital market for IV pain killers is about $1 billion, but is mostly generic. "There is nothing in the middle. We think we are the perfect in-between drug," Lu says.
Avenue is on track to initiate a Phase III program later this year, if it secures the financing it needs, after completing a pharmacokinetic study last year and holding an end of Phase II meeting with FDA.
Fortress is developing other drugs for the hospital market as well, including programs in development at Helocyte, which is developing treatments to prevent cytomegalovirus in patients undergoing allogeneic stem cell and solid organ transplants. CMV is asymptomatic in healthy individuals but can cause severe and life-threatening disease in those with weakened immune systems. Helocyte is developing two vaccines, PepVax and Triplex, that are engineered to induce a robust and durable virus-specific T-cell response to control CMV in transplant recipients.
The therapies were in-licensed from City of Hope and the laboratory of Don Diamond, PhD, chair of the Department of Experimental Therapeutics. The deal also involved an earlier-stage program Pentamer, a vaccine for the prevention of CMV transmission in utero.
Both PepVax and Triplex are in Phase II development in trials that are funded by grants from the National Cancer Institute. Grant funding is one path Fortress actively pursues to help finance drug development.
"The beauty of this company at this stage is we have got three novel biologics and we are heavily supplemented by grant funding," CEO Taffy says, adding that Helocyte has sufficient funding to complete the Phase II studies, at which point it would consider partnering or raising additional capital.
From hospital products to a pipeline of cancer drugs and potential treatments for rare diseases, Fortress has culled a pipeline of nearly 20 drugs. There is a lot to be excited about and the vision is ambitious. But executing on the bold strategy by bringing compelling, needed medicines to market is what will determine success.